Category: Late-Breaking General Abstract
4003 - VENETOCLAX TREATMENT OF REFRACTORY ITP IN CLL
Abstract Text:
Background: Chronic lymphocytic leukemia (CLL) is the most common adult leukemia. Approximately 2% of patients with CLL develop immune thrombocytopenic purpura (ITP) during the course of the disease. When resistant to steroids, this constitutes as indication for treatment of the CLL. Here we report of two patients with refractory ITP secondary to CLL successfully treated with venetoclax.
Methods: Patient data was taken from the patients and hospital records. Permission for off-label rituximab+venetoclax was obtained from Ministry of Health of Turkey.
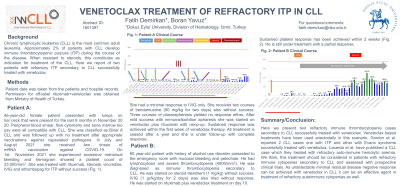
Results: Patient A: 46-year-old female patient presented with lumps on her neck that were present for the last 9 months in November 2020. Peripheral blood smear, flow cytometry and bone marrow biopsy were all compatible with CLL. She was classified as Binet A CLL and was followed up with no treatment after appropriate vaccinations against capsulated pathogens. In July and August 2021 she received two doses of mRNA vaccination against COVID-19. On 1st November 2021 she experienced excessive menstrual bleeding and hemogram showed a platelet count of 23.000/mm^3. She was treated with rituximab, steroids, vincristine, IVIG and eltrombopag for ITP without success (Fig. 1). She had a minimal response to IVIG only. She received two courses of bendamustine (90 mg/kg for two days) also without success. Three courses of plasmapheresis yielded no response either. After mild success with immunadsorbtion apharesis she was started on venetoclax plus rituximab with ramp-up. Sustained response was achieved within the first week of venetoclax therapy. All treatment is ceased after a year and she is under follow-up with complete response.
Patient B: 60 year-old patient with history of alcohol use disorder presented to the emergency room with mucosal bleeding and petechiae. He had lymphocytosis and severe thrombocytopenia (4000/mm3). He was diagnosed with immune thrombocytopenia secondary to CLL. He was started on steroid treatment (1 mg/kg) without success. IVIG (1 gr/kg/day for 2 days) was also tried without response. He was started on rituximab plus venetoclax treatment on day 10. Sustained platelet response has been achieved within 2 weeks (Fig. 2). He is still under treatment with a partial response.
Summary/
Conclusion: Here we present two reftactory immune thrombocytopenia cases secondary to CLL successfully treated with venetoclax. Venetoclax based treatments have been used anectodally in this scenario. Gordon et al. reported 2 CLL cases one with ITP and other with Evans syndrome successfully treated with venetoclax. Lacerda et al. have published a CLL case which they treated with refractory autoimmune hemolytic anemia. We think, this treatment should be considered in patients with refractory immune cytopenias secondary to CLL and assessed with prospective clinical trials. As undetectable minimal residual disease negative response can be achieved with venetoclax in CLL it can be an effective agent in treatment of refractory autoimmune cytopenias as well.
Keywords : immune thrombocytopenia, cll, venetoclax
Please indicate how this research was funded. : N/A
Please indicate the name of the funding organization.: N/A
Background: Chronic lymphocytic leukemia (CLL) is the most common adult leukemia. Approximately 2% of patients with CLL develop immune thrombocytopenic purpura (ITP) during the course of the disease. When resistant to steroids, this constitutes as indication for treatment of the CLL. Here we report of two patients with refractory ITP secondary to CLL successfully treated with venetoclax.
Methods: Patient data was taken from the patients and hospital records. Permission for off-label rituximab+venetoclax was obtained from Ministry of Health of Turkey.
Results: Patient A: 46-year-old female patient presented with lumps on her neck that were present for the last 9 months in November 2020. Peripheral blood smear, flow cytometry and bone marrow biopsy were all compatible with CLL. She was classified as Binet A CLL and was followed up with no treatment after appropriate vaccinations against capsulated pathogens. In July and August 2021 she received two doses of mRNA vaccination against COVID-19. On 1st November 2021 she experienced excessive menstrual bleeding and hemogram showed a platelet count of 23.000/mm^3. She was treated with rituximab, steroids, vincristine, IVIG and eltrombopag for ITP without success (Fig. 1). She had a minimal response to IVIG only. She received two courses of bendamustine (90 mg/kg for two days) also without success. Three courses of plasmapheresis yielded no response either. After mild success with immunadsorbtion apharesis she was started on venetoclax plus rituximab with ramp-up. Sustained response was achieved within the first week of venetoclax therapy. All treatment is ceased after a year and she is under follow-up with complete response.
Patient B: 60 year-old patient with history of alcohol use disorder presented to the emergency room with mucosal bleeding and petechiae. He had lymphocytosis and severe thrombocytopenia (4000/mm3). He was diagnosed with immune thrombocytopenia secondary to CLL. He was started on steroid treatment (1 mg/kg) without success. IVIG (1 gr/kg/day for 2 days) was also tried without response. He was started on rituximab plus venetoclax treatment on day 10. Sustained platelet response has been achieved within 2 weeks (Fig. 2). He is still under treatment with a partial response.
Summary/
Conclusion: Here we present two reftactory immune thrombocytopenia cases secondary to CLL successfully treated with venetoclax. Venetoclax based treatments have been used anectodally in this scenario. Gordon et al. reported 2 CLL cases one with ITP and other with Evans syndrome successfully treated with venetoclax. Lacerda et al. have published a CLL case which they treated with refractory autoimmune hemolytic anemia. We think, this treatment should be considered in patients with refractory immune cytopenias secondary to CLL and assessed with prospective clinical trials. As undetectable minimal residual disease negative response can be achieved with venetoclax in CLL it can be an effective agent in treatment of refractory autoimmune cytopenias as well.
Keywords : immune thrombocytopenia, cll, venetoclax
Please indicate how this research was funded. : N/A
Please indicate the name of the funding organization.: N/A
- FD
Fatih Demirkan, MD
Professor
Dokuz Eylul University, Division of Hematology
Izmir, Izmir, Turkey - BY
Boran Yavuz, MD
Fellow of Hematology
Dokuz Eylul University, Division of Hematology
Izmir, Izmir, Turkey

